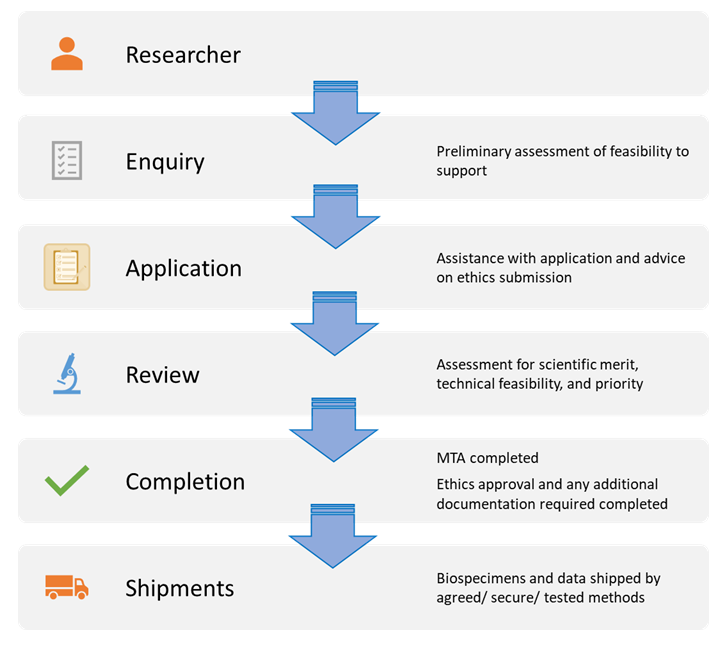
Details of the application process are provided here for academic researchers. (Industry researchers can apply for access through a similar but guided process)
Phase 1 – Review
1. Applicants contact us with the initial details of potential applications.
2. A teleconference meeting will be scheduled to discuss the project, to scope out general feasibility, provide advice on study design, and outline potential availability of relevant biospecimens and data.
3. Applicants who decide to proceed will then be provided with a Materials Access Application Form and advice on how to complete and submit this for review.
4. Our BBRS Access Committee will review the application and supporting documents for detailed biospecimen/data requirements using criteria outlined in CTRNet’s policy for ‘Material Release’.
5. Applications are rated as supportable, supportable with reservations, or unsupportable. Rationale for decisions is provided on request.
Phase 2- Release
If the application is rated as supportable the applicant must complete and provide the following:
1. A Material Transfer Agreement (provided to the applicant by the BBRS) that details the terms of use.
2. A certificate of Research Ethics Board approval from the applicant’s own institutional REB or appropriate independent REB documenting review and approval for the specific project proposed.
3. Acceptance of the final user fee and estimated shipping charges (provided to the applicant by the BBRS).
4. Acceptance of inclusion of project in our annual summary report of research projects. This list is provided to the UBC BC Cancer REB as part of our annual renewal application and also to funding agencies.
For some applications, at our discretion, the applicant may also be asked for additional materials including:
1. Documentation of adequate experience in handling human biospecimens and data. This can also be fulfilled by completion of a short online education module on handling biospecimens and biobanking processes. This Researcher Education Module can be accessed here and can be completed by the most relevant individual involved in conducting the research project (e.g. graduate student, post-doctoral fellow, technologist).
2. Documentation of funding for the specific project.
3. Agreement around timing of shipping study materials in several test and main study batches.
4. Confirmation of receipt of a small test shipment of biospecimens to confirm delivery processes and routing.
5. Confirmation and provision of experimental evidence of successful performance of the relevant assay(s) on our material in this test batch.